Book Leflunomide Metabolite Appointment Online Near me at the best price in Delhi/NCR from Ganesh Diagnostic. NABL & NABH Accredited Diagnostic centre and Pathology lab in Delhi offering a wide range of Radiology & Pathology tests. Get Free Ambulance & Free Home Sample collection. 24X7 Hour Open. Call Now at 011-47-444-444 to Book your Leflunomide Metabolite at 50% Discount.
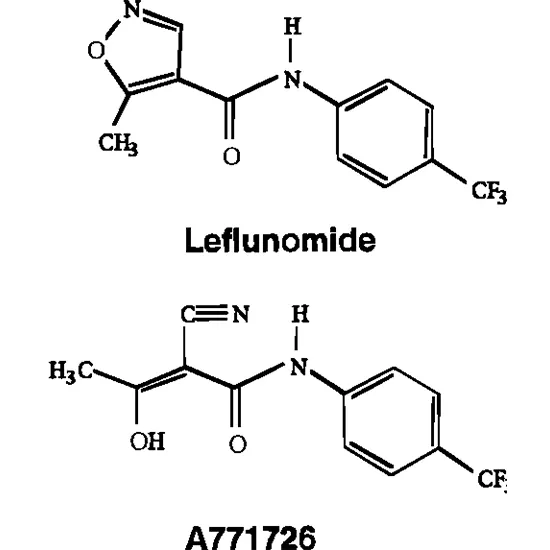
Rheumatoid arthritis is treated with the medication leflunomide (Arava®). It works by blocking the body from creating the antibodies that cause the disease's symptoms. (inflammation, edema, stiffness, joint pain). Leflunomide is a prodrug, meaning that it is given to the patient in an inactive form that the liver then transforms into an active metabolite.
Leflunomide Metabolite: The immunosuppressant drug leflunomide is used to treat rheumatoid arthritis. Teriflunomide, the drug's active form, is quickly and thoroughly metabolized from leflunomide. Patients receiving the indicated daily dosages experienced steady state plasma values of up to 63,000 ng/mL. All women of reproductive potential who stop taking leflunomide and those who are getting treatment but want to get pregnant are advised to go through the cholestyramine medication elimination process. This approach includes two independent tests that are at least 14 days apart to confirm that the plasma concentration of teriflunomide is less than 20 ng/mL.
Methodology Mass spectrometry and chromatography
The characteristics of this test's analytical performance have been established by Quest Diagnostics. The FDA has not endorsed or cleared it. This test is used for clinical applications and has been validated in accordance with CLIA requirements.
Patients who were taking daily doses of 5, 10, or 25 mg of leflunomide had mean steady state plasma concentrations of teriflunomide of 8,800, 18,000, and 63,000 ng/mL, respectively. It is advised that pregnant women who stop taking leflunomide treatment go through the cholestyramine medication elimination process. This approach involves confirming, through two distinct tests done at least 14 days apart, that the plasma concentration of teriflunomide is less than 20 ng/mL.
Plasma EDTA in serum
Specimen Transport
Sample preparation in a laboratory centrifuge and storage in a cold room with a toxicology box.
Time Required for Sample Preparation in a Centrifuge: 3 working days
4 oC Sample Stability
The specimen for this test must be obtained, processed, aliquoted, and refrigerated within 45–60 minutes of the specimen being obtained. Patients must be sent to a Dynacare Laboratory and Health Services Center for collection if they are unable to comply with these requirements. Never employ a gel barrier tube. Transfer serum to a transport tube after centrifuging it.
Transport and Storage: Keep chilled and ship chilled.
To ensure proper dosing, this test is intended for the therapeutic medication monitoring of teriflunomide/leflunomide levels. This test is not intended to track the disappearance of teriflunomide in teriflunomide-free women who are capable of having children. Eurofins provides access to this service. (Sanofi-Aventis kit)
| Test Type | Leflunomide Metabolite |
| Includes | Leflunomide Metabolite (Pathology Test) |
| Preparation | |
| Reporting | Within 24 hours* |
| Test Price |
₹ 5400
|

Early check ups are always better than delayed ones. Safety, precaution & care is depicted from the several health checkups. Here, we present simple & comprehensive health packages for any kind of testing to ensure the early prescribed treatment to safeguard your health.