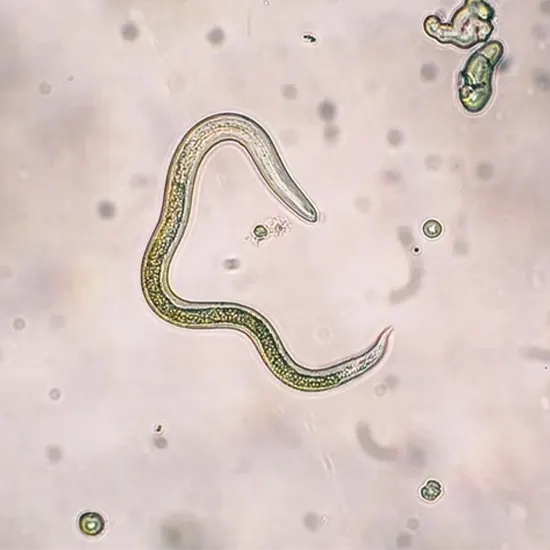
Toxocariasis is the clinical term used for infection of human hosts by Toxocara canis or Toxocara cati. Both are roundworm nematodes, in order of roundworm, superfamily Ascaridae, and Ascaridae. Their definitive hosts are...
Toxocariasis Also known as visceral larva migrans [VLM]. Toxocariasis is the clinical term used for infection of human hosts by Toxocara canis or Toxocara cati. Both are roundworm nematodes, in order of roundworm, superfamily Ascaridae, and Ascaridae. Their definitive hosts are domestic dogs and cats, and as adults they live in the lumen of the small intestine.
Infection can occur when the host ingests viable, embryonated eggs from contaminated sources (such as soil or earthworms). Infection may also be acquired in utero (i.e., transplacental) from an infected mother if she picks up more infective eggs. In contrast, the human host is unusual in terms of life cycle completion.
Infectious larvae hatch after ingesting the eggs, but the larval stage does not develop into mature adults. Instead, they wander through the body for months to years, damaging the tissues they happen to invade.The ability of eukaryotic parasites to survive for such long periods in mammals is unusual. Few others have developed long-term survival strategies.
That is, the adult stage of schistosomes is 10 to 25 years, her 1st instar larvae of trichinella live the lifespan of the host, adult phytonematodes of some species live 10 to 15 years, and most tapeworms live 10 to 15 years. species live in the larval stage.
Live from 5 to 10 years. To accomplish this daunting feat, these parasites have all evolved unique mechanisms to evade the host's immune system. Toxocara is no exception.
There is visceral larval migration (VLM), which involves disease involving major organs, and ocular larval migration (OLM), in which the pathological effects of toxocariasis are limited to the eye and optic nerve.
History of Discovery
Human infection with Toxocara sp. It was first described by Wilder in 1950. He identified larvae of an unknown species of nematode in a child's retinal granuloma. In 1952, Beaver and his colleagues reported a similar series of children who presented with severe eosinophilia and suffered from severe long-term multisystem disease.
From this group of patients, they described most of the clinical features of his VLM and identified the pathogen in histopathological sections of tissue obtained at biopsy as T. canis larvae or T. Cat.
Since that time, larvae of these two parasite species have been identified in various eye lesions and throughout the body in patients from all regions of the world. Today, the entire public health community recognizes that toxocariasis in all its clinical forms poses significant health risks, especially to children with pica.
Life Cycle
Ingestion of embryonated eggs of TToxocar initiates infection in both definitive and mutant hosts. Children may come in contact accidentally while playing in sandboxes or playgrounds contaminated with Toxocara eggs. This situation results from indiscriminate defecation in these areas by cats and dogs harboring adult worms. After ingestion, the eggs hatch and release larvae (juveniles).
The larvae have the opportunity to penetrate the small intestine, enter the circulatory system and move freely throughout the body to invade all organs. It is unknown whether this is a second or third instar larva.
In the final host, the young parasite completes a life cycle that in many respects resembles that of the roundworm that infects humans. Transplacental infections are common in both dogs and cats.
In addition, the definitive host can be infected by ingesting embryonated eggs carried by paratene hosts such as earthworms, ants, and other soil-dwelling invertebrates. Humans can develop unusual infections by ingesting the same animals.
Worms mature in the small intestine approximately 60 to 90 days after the larvae hatch. Mating then occurs and an embryonated egg is produced, which is excreted in the faeces. Embryogenesis occurs in the soil approximately one week after deposition. The longer incubation times are due to the lower temperatures. In northern latitudes, eggs remain dormant until warmer temperatures in spring, causing embryogenesis.
Clinical Aspect
The extent of host damage and associated signs and symptoms induced depends on which tissue is invaded. The central nervous system, including the liver, lungs, and eyes, appears to be the most sensitive.
Additionally, the number of pups to move and the age of the host are two important factors in determining whether a particular individual's condition is beyond the clinical horizon.
The pathological outcome largely depends on the adolescent's death. Her death marks the development of marked late-type and immediate-type hypersensitivity reactions. Inflammation manifests as an eosinophilic granuloma.
In the eye, her migratory 3rd instar larvae can damage the retina, causing a granulomatous reaction and causing visual impairment. In severe cases, granulomas are responsible for vision loss. These pathological symptoms have been misdiagnosed as retinoblastoma in the past. Today, with reliable immunodiagnostic reagents and methods, OLM is rarely confused with other clinical units.
Epidemiological evidence suggests that ocular disease usually occurs without systemic involvement and vice versa, and he Culture Aerobic Ocular Fluid referred to her two manifestations of this infection as OLM and VLM. It leads to a proposal to classify. T lineage may exist.
CLINICAL SIGNS AND SYMPTOMS
VLM
VLM is primarily a disease of young children (under 5 years of age). accompanied by fever. enlarged and necrotic liver; enlarged spleen; CBC test lower respiratory tract symptoms (particularly asthma-like bronchospasm); eosinophilia, sometimes approaching 70%.
Hypergammaglobulinemia of immunoglobulin M (IgM), IgG and IgE classes. In the latter of these cases, x-ray levels of IgE/anti-IgE immune complexes are elevated and symptoms become more pronounced. Myocarditis, nephritis, and CNS involvement have been reported.
Central nervous system involvement can result in seizures, neuropsychiatric symptoms, or encephalopathy. It is increasingly recognized that more subtle clinical manifestations may also occur as a result of long-term exposure to moving puppies.
So-called latent toxocariasis span the spectrum from asymptomatic infections to larvae that migrate to specific target organs. In the CECT Chest lung, migration of larvae can lead to asthma. T. canis has been suggested as an environmental risk factor for asthma in some urban populations.
Similarly, T. canis in the brain has been implicated as one of the causes of so-called idiopathic seizure disorders and one of the causes of functional bowel disorders.
One study showed that Toxocara contributes to at least two types of skin disease: prurigo and urticaria. Echocardiography Experimental infection of mice impairs learning and memory, both of which appear to be dose- and time-dependent. It is therefore reasonable to speculate that similar phenomena are likely at work in long-term human infections as well.
OLM
OLM usually occurs in children aged 5 to her 10 years and typically presents as unilateral visual impairment, sometimes accompanied by strabismus. The most serious consequence of infection is retinal infiltration, leading to Ultrasound granuloma formation. It usually occurs at the peripheral or posterior pole. These granulomas pull on the retina, causing macular distortion, astigmatism, or detachment.
The degree of visual impairment depends on the area affected, and blindness is common. OLM can also cause diffuse endophthalmitis or papillitis. Secondary glaucoma may follow. In at least one rare case, a choroidal neovascular membrane formed after prolonged infection with Toxocara, previously manifesting as choroiditis.
Diagnosis
VLM should be suspected in pediatric patients with febrile illness of unknown cause and eosinophilia. A history of hepatosplenomegaly and evidence of multiple system disease and pica make the X Ray Ocular Region diagnosis of VLM more likely. Similarly, OLM should be suspected in children with unilateral vision loss and strabismus.
Diagnostic tests for VLM are primarily immunological tests. The Precipitin test is subject to cross-reactivity with common larval and blood group substance A antigens. The enzyme-linked immunosorbent assay (ELISA), which uses antigens shed by second instar larvae, is sufficiently specific to be the best indirect test.
Tests for diagnosis of this infection. Recombinant antigen was generated from second stage larvae of T. canis. This promises even higher specificity for high-confidence tests using ELISA (approximately 92%). The ELISA also has a very high sensitivity (approximately 78%) with titers >1.
Other signs of infection include hypergammaglobulinemia and elevated isohemagglutinin levels. Therefore, the above clinical disease cluster, pica history, eosinophilia, and positive serology are strongly indicative of the diagnosis.
OLM is diagnosed primarily based on clinical criteria during an ophthalmologic examination. Immunodiagnostic tests used for VLM are not reliable for OLM. In one study, only 45% of patients clinically diagnosed with OLM had titers >1.
Treatment
Albendazole is a drug used to treat toxocariasis. The patient who received her 5-day treatment with albendazole (10 mg/kg body weight/day in two doses) improved compared with those treated with the older anthelmintic, thiabendazole.
Albendazole 400 mg twice daily for 5 days is the current recommended regimen. The other commonly used benzimidazole, mebendazole, is poorly absorbed outside the gastrointestinal tract, so this drug is second-line therapy, but in 21 days he reported some success in patients who ingested more than 1 g. It has been.
Symptomatic treatment, including administration of corticosteroids, has helped control severe allergic symptoms due to infection. OLM is treated with surgery (vitrectomy), anthelmintic chemotherapy, and/or corticosteroids.









