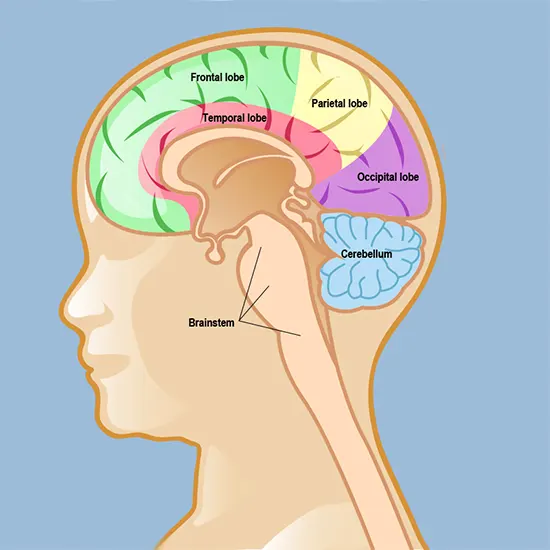
Astrocytoma form in astrocytes, a type of star-shaped glial cell in the cerebrum. This is the most common glioma and usually affects the brain and sometimes the spinal cord. Glial tumors account for 60% of brain tumors. They...
Introduction
Astrocytoma form in astrocytes, a type of star-shaped glial cell in the cerebrum. This is the most common glioma and usually affects the brain and sometimes the spinal cord. Glial tumors account for 60% of brain tumors. They are a common cause of mortality and morbidity in both young and old.
Etiology
The underlying cause of most primary brain tumors has not been identified, and the only established risk factor is exposure to ionizing radiation. No evidence of association with other factors, such as exposure to electromagnetic fields (mobile phones), head injuries, or occupational risk factors.
Children who receive prophylactic radiotherapy for acute lymphoblastic leukemia (ALL) may have a 22-fold increased risk of developing central nervous system malignancies within approximately 5 to 10 years. Radiation therapy for pituitary adenomas has been shown to increase the risk of developing glioma by 16 times. Diseases such as Turcot syndrome, p53 mutation (Li-Fraumeni), and NF1 syndrome have a genetic susceptibility to develop gliomas. Approximately 66 of the low-grade astrocytoma have p53 mutations.
Epidemiology
The age-standardized incidence of glioma was 4.7/100,000 person-years.
Mortality
A typical survival area is:
- WHO Grade I (pilocytic astrocytoma) - >10 years.
- WHO grade II (low-grade diffuse astrocytoma) - >5 years.
- WHO grade III (anaplastic astrocytoma) - about 2-5 years.
Minimal racial differences were observed.
Gender
There is no gender difference in pilocytic astrocytomas. I have one of the low grades astrocytomas. In anaplastic astrocytomas, males are one of hers with a significant male predominance.
Year
The chance of pilocytic astrocytoma increases in the first 20 years. In the 30- to 40-year-old group, low-grade astrocytomas predominate, accounting for about a quarter of adult cases. The distribution of low-grade astrocytomas is as follows:
- Ten%:
less than 20 years.
- 60%:
20-45 years old.
- 30%:
45+ age.
The average age for anaplastic astrocytoma is about 40 years.
Pathophysiology
The local effects of astrocytoma are the result of multiple mechanisms. These include direct entry and competition for oxygen, resulting in hypoxic damage to normal brain parenchyma. In addition, free radicals, neurotransmitters, and inflammatory mediators also contribute to homeostasis. The mass effect caused by tumors is also responsible for various clinical signs and symptoms.
Histopathology
- Common Types Pilocytic Astrocytoma.
- It is benign in nature.
- Has cystic consistency.
- Location is mostly infratentorial.
- Presents classically in childhood.
- Can be resected surgically.
Variant:
Grade I
- Pilomyxoid astrocytoma.
- Diffuse Astrocytoma.
Grade II
- Usually seen in adults.
- May progress to glioblastoma.
- Anaplastic Astrocytoma.
Grade III
- Also usually seen in adults.
- A lack of endothelium proliferation is present.
- Commonest malignant brain tumor.
- Peak age is around 65 years.
- Very bad prognostication.
Variants:
Giant cell glioblastoma, Gliosarcoma.
Pleomorphic Xanthoastrocytoma (PXA).
The prognosis is usually good.
Age group affected:
Children & adults (young).
- Has big lipidized cells.
- May mimic malignant tumor.
- Subependymal Giant Cell Astrocytoma (SEGCA).
- Located mostly intraventricularly.
- Benign in nature.
- Affects the adolescent age group.
- Associated with Tuberous sclerosis.
- Gliomatosis Cerebri.
Presence:
1. Type 1 GC:
No IDH 1/2 mutation, diffusely growing.
2. Type 2 GC:
IDH 1 mutation +, solid component present.
High-grade
- Location: Spina cord, brainstem, and thalamus in the middle.
- Adolescents & children are mostly involved.
- DPIG (Diffuse pontine intrinsic glioma) is included.
- New entry after 2016 WHO classification.
Poor prognosis is usually there when:
- Absence of EGFR amplification.
- Presence of unmethylated MGMT promoter.
Enhancement in MRI:
May/may not be present.
Histologically, they might be anything from barely hypercellular to fully developed glioblastoma.
Gliosarcoma
- It is regarded by the WHO as a glioblastoma variety.
- With about 200 reported cases, it is exceedingly uncommon.
- Contains elements of sarcoma and glioblastoma (fibroblastic, osseous, cartilage, smooth & striated muscle, fat cells).
- It commonly develops in the temporal lobe and resembles glioblastomas.
- Prognostically like glioblastoma.
Gliofibroma
- It is very rare.
- Usually found in children.
- Has a fibroblastic part, which is non-malignant.
History and Physical
History
There are two types of symptoms: general and localized.
Common general symptoms include early-morning headaches, nausea, vomiting, problems with cognition, personality changes, and gait problems. Errors in the visual field, aphasia, or seizures are examples of localizing symptoms. A visual field deficiency is frequently not detected by the patient and may only be discovered after it causes harm, like in car accidents.
Seizures occur in about 90% of patients with low-grade glioma.
Physically
Always do a complete neurological examination. All signs are localized and should be easily identifiable. Similarly, sensory level delineation can help identify malignant levels in the spinal cord. Cranial nerve palsy is a good sign of intracranial localization. Neuroimaging should be done to further confirm the location of the tumor.
Evaluation
Neuroimaging is the only test required to diagnose brain tumors. Whenever possible, gadolinium-enhanced MRI imaging should be used. If there are contraindications for MRI, such as B. In situ joint implants or pacemakers, computed tomography or CT can be performed.
MRI (fluid-attenuated reversal recovery) of the FLAIR sequence is performed because low-grade gliomas have no contrast enhancement. If a tumor is found, a neurosurgeon should do a biopsy. It simply removes a small amount of tumor tissue and sends it to a neuropathologist for examination and classification. His CT images of low-grade astrocytomas are also generally equivocal.
They present as homogeneous, poorly defined, poorly defined, nonenhancing lesions. Contrast may be enhanced in contrast-enhancing astrocytomas. Metastatic disease is possible. Therefore, whole-body imaging should also be considered to search for alternative primaries.
T2 hyperintensity is seen in astrocytomas on MRI, while isointensity is present in T1. Tumor vascularity is very important. Therefore, new techniques are being developed to identify it. Functional MRI is an emerging imaging modality. It makes sense to demarcate different brain regions according to function preoperatively.
These can provide information about tumor metabolic processes, blood flow characteristics, and tumor composition. It can be used to determine whether lesions are progressive or necrotic after chemotherapy or radiation therapy.
Treatment / Management
The WHO classification (2016) divides diffuse astrocytomas into grades II, III, and IV. Invasive grade I astrocytomas were not mentioned. It is based on the following four qualities:
1. Nuclear atypia:
Nuclear pleomorphism and hyperchromasia.
2. Mitosis:
need to clarify.
The Ki67 proliferation index is used to distinguish grade II tumors from grade III tumors.
3. Microvascular proliferation:
In lower-grade gliomas (such pilocytic astrocytoma), the globeruloid form is more prevalent but has a less severe prognosis. Endothelial proliferation is detected in the lumen of big vessels. It is uncommon and more frequently associated with high-grade gliomas.
4. Necrosis:
Coagulative necrosis.
Pseudopalisading necrosis.
WHO Histological Grading:
Grade I (diffuse Astrocytoma).
Grade II (diffuse astrocytoma):
Nuclear atypia alone.
Grade III (anaplastic astrocytoma):
Nuclear atypia + focal/dispersed anaplasia.
Grade IV (glioblastoma):
Microvascular proliferation, necrosis, nuclear atypia, or mitosis.
Pilocytic astrocytoma:
Corresponding to WHO-grade I
SEGCA (subependymal giant cell astrocytoma):
Grade I
Pleomorphic xanthoastrocytoma.
Grade II and anaplastic pleomorphic.
Grade III
Glioma Treatment Recommendations Based on Grade.
Grade I uncommon, typically noninvasive and are considered benign. microvascular proliferation, necrosis, nuclear atypia, or mitosis.
Surgery has the potential to cure this condition, however if total surgical excision is not possible, expectant care or radiotherapy are utilized instead.
Grass II (low-grade infiltrative astrocytoma, oligodendroglioma, mixed gliomas).
Surgery is advised with the safest resection possible.
age >40 years, size 6 cm, crossing midline, and presence of neurologic deficit prior to resection are unfavorable prognostic variables. Three or more factors increase risk.
Low-risk, < 40-year patients.
Standard of care:
Surgical resection followed with EBRT (60 Gy in 30 to 35 fractions) and adjuvant temozolomide, 75 mg/m2/day orally on days 1 to 42, typically one to one and a half hours before radiation.
Post–radiation therapy:
Continue temozolomide at higher doses of 150 to 200 mg/m^2/day PO on days 1 to 5 every 28 days or PCV (procarbazine, lomustine, vincristine):
- Lomustine (CCNU) 90–130 mg/m2 PO on day 1, procarbazine 60–75 mg/m2 PO on days 8–21, and vincristine 1.4 mg/m2 IV (maximum 2 mg/dose) on days 8–29; administer every six weeks for up to four cycles with delayed irradiation.
Grade IV (glioblastoma):
Standard treatment consists of surgical resection followed by EBRT (60 Gy in 30–35 fractions) and temozolomide 75 mg/m^2/d orally on days 1–42, usually 1–1.5 hours before radiation. Day adjunctive therapy.
After irradiation:
Continue temozolomide at high doses of 150–200 mg/m^2/day and administer her P
O on days 1–5 every 28 days.
Recommendations for recurrent tumors:
Reoperation, carmustine wafers, and alternative chemotherapy regimens. Reirradiation is rarely helpful. Bevacizumab, a humanized VEGF monoclonal antibody, is effective in recurrent glioblastoma, prolonging progression-free survival and reducing peritumoral edema and glucocorticoid use.
Approach method:
As with other malignancies, multimodal therapeutic approaches are being pursued that include surgical, medical, and radiation-oncological treatments.
Low-grade astrocytoma:
These low-grade tumors are very slow-growing, raising questions about the risk-benefit ratio of surgery. A study by Ishkanian et al. However, overall survival was the same. Astrocytoma grade 2:
Radiation therapy and adjuvant chemotherapy are superior to radiation therapy alone. One study showed that chemotherapy with vincristine, procarbazine, and lomustine improved 10-year PFS after radiation therapy (51% vs. 21%).
Anaplastic astrocytoma:
Multimodality therapy is integrated, including surgery, radiation, and chemotherapy (adjuvant temozolomide). Data on the combination of temozolomide is lacking, but some studies have shown improved survival (46% vs. 29%). IDH (isocitrate dehydrogenase) mutations are also associated with improved 5-year survival (79% vs. 22%).
Anaplastic astrocytoma responds better to chemotherapy than glioblastomas. For relapses, temozolomide responds better. Recurrent tumors treated with nitrosoureas showed approximately 35% response to temozolomide. Furthermore, 6-month survival was also favorable (46% vs. 31%). The adjuvant carmustine also showed a small survival advantage.
Prophylactic initiation of antiepileptic drugs is controversial. People who complain of seizures should start with antiepileptic drugs. For attacks, patients usually started on levetiracetam, topiramate, lamotrigine, valproic acid, and lacosamide.
These medications have fewer negative effects on the hepatic microsomal enzyme system. Other drugs, such as phenytoin and carbamazepine, are less commonly used because they are potent enzyme inducers that can interfere with the metabolism of glucocorticoids and chemotherapy drugs.
Corticosteroids are also anti-inflammatory. It is used to provide superior symptom relief due to its effectiveness. This leads to a reduction in the mass effect. Dexamethasone is the glucocorticoid of choice due to its low mineralocorticoid activity. The initial dose is usually 12 to 16 mg/day orally or intravenously in divided doses. (Both are equivalent). Concomitant prophylaxis of gastrointestinal ulcers should be prescribed along with the administration of corticosteroids.
Surgical Care
Surgery is used to remove/shrink tumors. In addition, tissue biopsy by the surgeon enables histological diagnosis. Other symptom relief measures include relief of procedures such as ICT (intracranial tension), VP shunts, and EVD insertion. 8.8 months). Total superior resection (i.e., removal of tissue beyond MRI-defined abnormalities) is also recommended for low-grade gliomas, suggesting that this strategy may prolong overall survival.
Overview of other drugs
Venous thromboembolic disease occurs in 20% to 30% of patients with high-grade gliomas and brain metastases. Therefore, prophylactic anticoagulants should be used in hospitalized and non-ambulatory patients. Therapeutic doses of anticoagulants can be administered without raising the risk of bleeding into the tumour to those with deep vein thrombosis (DVT) or pulmonary embolism.
Antineoplastic agent
Temozolomide (alkylating agent):
An oral alkylating agent that converts to MTIC at physiological pH. 100% bioavailable. About 35% cross the blood-brain barrier.
mTOR inhibitors have also been suggested for the treatment of grade 1 astrocytomas.
Differential diagnosis
- Glioblastoma multiforme.
- Brain metastasis.
- Brain abscess.
- Oligodendroglioma.
- Encephalitis.
- Multiple sclerosis.
- Primary CNS lymphoma.
- Toxoplasmosis.
Staging
Astrocytomas are not staged. As previously mentioned, only prognostic determinants grading is performed.
Prognosis
It favors low-grade tumors, with survival approaching 7-8 years after surgery. In anaplastic astrocytoma, treatment focuses on improving symptoms. Radiation therapy of partially resected tumors increases survival after surgery. Survival after postoperative radiation is nearly double that of pure surgical intervention (5 vs. 2.2 years).
Genetics also plays a role in determining prognosis for certain types of tumors. Oligodendrogliomas with alterations in Ch 1p19q respond better to PCV [procarbazine, CCNU (lomustine), and vincristine]. Genetics is an emerging field of personalized tumor therapy.
Kallikrein levels are associated with patient prognosis.
Complications
Neurosurgical interventions always carry the risk of surgical complications. Complications of chemotherapy and radiotherapy are observed as well as cancer treatments, and effective drugs are being developed to adequately treat these complications.